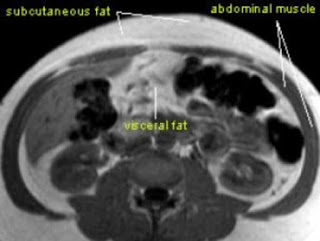
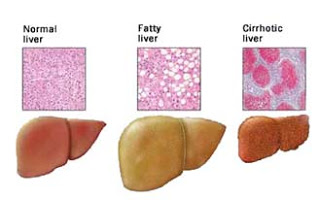
It has become clear that bariatric (obesity) surgery can result in substantial improvement, or even remission, of type 2 diabetes for some people. Two new articles from the New England Journal of Medicine now add to our knowledge on this topic.
(For the non-scientists in the audience, feel free to skip down to ‘So what does this mean?‘ below.)
One of these studies, by Mingrone and colleagues, looked at 60 patients randomized to receive either gastric bypass surgery, biliopancreatic diversion surgery (BPD), versus their usual diabetes care with medications, and examined how many people would be in remission from their diabetes 2 years later. They found that 75% of the patients who had gastric bypass and 95% of the patients who had BPD were in remission, whereas none of the control group was in remission. Interestingly, none of age, gender, baseline body mass index, nor duration of diabetes were predictive of remission.
The other study, by Schauer and colleagues, randomized 150 patients to receiving either gastric bypass surgery, sleeve gastrectomy, versus usual medical care of type 2 diabetes, with the goal being to see how many patients from each group could achieve very tight control of their diabetes (defined by A1C of 6% or less) at one year. They found that more patients who had surgery achieved this goal (42% of gastric bypass patients and 37% in sleeve gastrectomy patients), compared with 12% of patients receiving medications alone. At one year, the mean A1C in the medication group was 7.5%, compared to 6.4% in the gastric bypass group and 6.6% in the sleeve gastrectomy group.
While each of these studies could be discussed with chapters of detail, for purposes of brevity I’ll highlight just a couple of important caveats. While a strength of these studies is that they are randomized clinical trials (very hard to do in the area of bariatrics), both studies are small. In the Mingrone study, BPD was used as a surgical technique, which is a fairly drastic surgery (it bypasses more of the bowel than gastric bypass), and is currently only experimental. The longer term follow up of these patients is important, as other studies now suggest that at 5 years after bariatric surgery, about half of the diabetes cases that initially went into remission come back (though the diabetes-free years are undoubtedly still of substantial health benefit). In the Schauer study, one could argue that the diabetes control goal (A1C 6% or less) was too tight and not appropriate for routine clinical care, given that we no longer strive for this tight control in most cases of type 2 diabetes because of the potential risk of harm (see the ACCORD trial). What is interesting to me, however, is that the overall control was better in the surgical groups compared to the control group.
So what does this mean? These studies show us that bariatric (obesity) surgery can put type 2 diabetes into remission, and can improve control of diabetes in those who don’t go into remission. However, it must be noted that remission does NOT mean cure – each patient must be followed on a lifelong basis and monitored for possible recurrence of diabetes down the road. These surgeries have significant risk associated with them, and the balance of benefit versus risk has to be considered on a patient-by-patient basis. The improvement in diabetes does not appear to depend on how much the person weighs before surgery, implying that the current body mass index (BMI) critieria for selecting patients for surgery may not be the right way to determine who would benefit the most. (More research needs to be done to figure out what does predict best success with bariatric surgery.)
Overall, (and as noted in the accompanying editorial), studies such as these suggest that bariatric surgery should perhaps not be a ‘last resort’ in the treatment of patients with obesity and type 2 diabetes.
Dr Sue Pedersen www.drsue.ca © 2012 drsuetalks@gmail.com
Follow me on Twitter for daily tips! @drsuepedersen