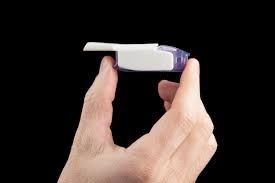
In an effort to find alternative ways to administer insulin apart from the current standard of injecting it under the skin, a new inhaled insulin has recently been made available in the United States.
Called Afrezza, the inhaled insulin is taken at mealtime, and is used instead of fast-acting insulin injection at mealtimes (it does not replace the need for long acting insulin in those who need it). It is rapidly absorbed by the lungs, has its peak action at under an hour, and is gone in 2.5 to 3 hours (a little faster than the fast acting mealtime injectable insulins currently available).
While clinical trials have been done, showing that the inhaled insulin is comparable in efficacy to mealtime injectable insulin in type 1 and type 2 diabetics, these studies have been criticized in that diabetes control in these studies was not as tight as recommended by diabetes guidelines (ie, hemoglobin A1C of 7% or less).
There are a number of additional concerns with inhaled insulin, including:
1. Dose increments are large (it has to be adjusted by 4 unit increments, whereas injectable insulin can be adjusted by as little as 0.5 unit increments). This makes it harder to fine-tune blood sugar control.
2. Inhaled insulin cannot be used in smokers or people with lung disease (such as asthma).
3. Inhaled insulin causes a small decrease in lung capacity (forced expiratory volume). Lung function has to be monitored in people using inhaled insulin.
My take: Due to the concerns above, I don’t see this as a game changer in diabetes care. Should this inhaled insulin become available in Canada, I will continue to recommend the currently available injectable mealtime insulins to my patients. That being said, for people who are very resistant to the idea of taking an injectable mealtime treatment, this could offer an (inferior) alternative way to give insulin to those people – ie, it may be a better alternative to no mealtime insulin at all in people who need it.